For Research Use Only. Not for human or veterinary use.
Ambuic AcidProduct ID: BLK-1110 | |
| Structure | 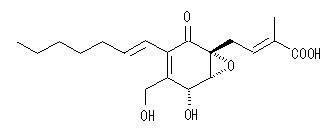 |
| CAS | 340774-69-8 |
| Molecular Formula | C19H26O6 |
| Molecular Weight | 350.41 |
| Price | 1mg: USD 180.00 / 5mg: - / 25mg: - |
| Storage | -20°C |
| Solubility | 1mg/ml ethanol, methanol, DMSO |
| Purity | >95% |
| Source Organism | Pestalotipsis sp. |
| Summary | Ambuic Acid Fungal-derived quorum sensing inhibitor that blocks AIP biosynthesis in Gram-positive bacteria and is studied in antivirulence research. |
| Details | |
| Mechanism | Inhibits Gram-positive quorum sensing by suppressing autoinducing peptide biosynthesis (agr/fsr systems), reducing virulence gene expression without affecting bacterial growth. |
| Primary Target | agr quorum sensing system; fsr quorum sensing system; Autoinducing peptide (AIP) biosynthesis machinery |
| Pathway | Quorum sensing pathway; Bacterial virulence regulation |
| Chemical Class | Epoxidized cyclohexenone polyketide |
| Discovery Year | 2009 |
| Keywords | quorum sensing inhibitoragr system inhibitorfsr system inhibitorautoinducing peptide biosynthesis inhibitoranti-virulence agentepoxide-containing polyketidefungal metabolitegram-positive bacteria research tool |
| References |
|
Documents | |
| ← Back to Catalog | |